Pyruvate Kinase
Carbohydrate Metabolism
Pyruvate kinase catalyzes the final step in glycolysis, converting phosphoenolpyruvate to pyruvate.
Pyruvate kinase catalyzes the final step in glycolysis, converting phosphoenolpyruvate (PEP) to pyruvate, while generating ATP in the process. This reaction is one of the key regulatory points in glycolysis, as it determines the rate at which glucose is metabolized for energy production. Pyruvate kinase is a tetrameric enzyme, and its activity is tightly regulated by various allosteric and covalent mechanisms to coordinate cellular energy needs.
The conversion of PEP to pyruvate is energetically favorable, as it leads to the generation of ATP. This step is essential for the continued operation of glycolysis, providing energy for cells under anaerobic conditions (when oxygen is scarce) or during periods of rapid energy demand, such as in muscle cells during exercise.
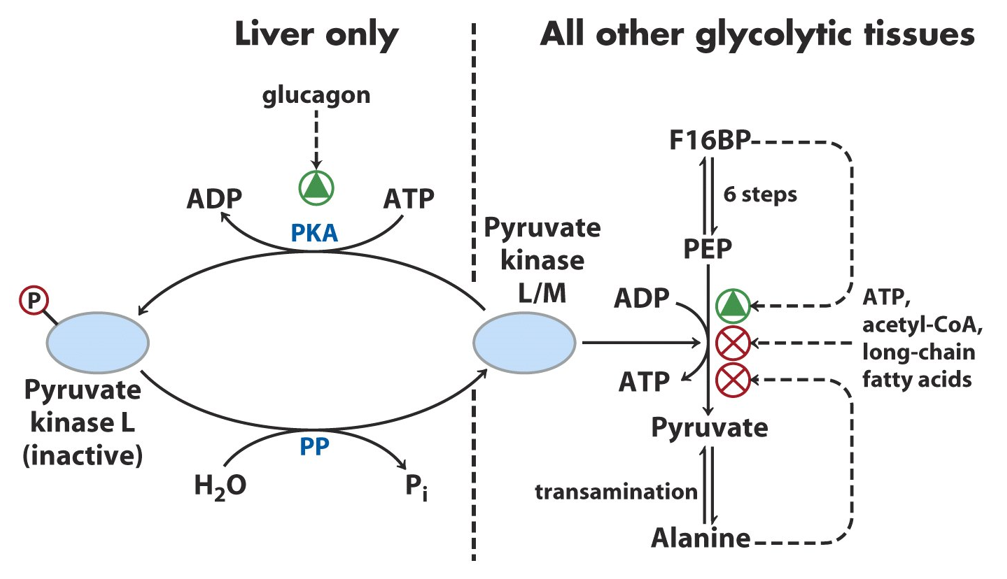
Pyruvate kinase is regulated through both allosteric regulation and covalent modification. The enzyme exists in different isoforms, with the most commonly studied being the M-type (muscle) and the L-type (liver). In muscle cells, pyruvate kinase activity is allosterically activated by fructose-1,6-bisphosphate, an intermediate earlier in the glycolytic pathway, reflecting the need for continued glucose breakdown when glycolysis is active. This positive feedback ensures that when glycolysis is proceeding rapidly, pyruvate kinase is upregulated to continue ATP production.
In contrast, pyruvate kinase in the liver is subject to covalent modification via phosphorylation. When glucose levels are high, insulin signaling promotes the dephosphorylation and activation of pyruvate kinase, facilitating glycolysis and promoting the storage of excess glucose. On the other hand, when glucose levels are low (as during fasting or in response to glucagon signaling), pyruvate kinase is phosphorylated by protein kinase A (PKA), leading to its inactivation and a reduction in glycolytic flux.
Additionally, ATP and alanine act as allosteric inhibitors of pyruvate kinase, signaling that the cell has sufficient energy and that glycolysis should slow down. In the liver, acetyl-CoA also inhibits pyruvate kinase, providing a regulatory link to fatty acid metabolism, as high levels of acetyl-CoA indicate that fat stores are abundant and glucose metabolism should be downregulated.
Through these various mechanisms, pyruvate kinase helps integrate metabolic signals from glucose, fatty acids, and overall cellular energy status, ensuring that glycolysis and energy production are appropriately regulated.


