Pyruvate Dehydrogenase Complex
Carbohydrate Metabolism
Pyruvate dehydrogenase complex converts pyruvate into acetyl-CoA, linking glycolysis and the citric acid cycle.
Located in the mitochondria, this large enzyme complex plays a pivotal role in aerobic metabolism by facilitating the decarboxylation of pyruvate, produced from glucose during glycolysis, to form acetyl-CoA. This acetyl-CoA is then utilized in the citric acid cycle to generate high-energy molecules like NADH, FADH2, and ATP, essential for cellular function.
The pyruvate dehydrogenase complex is composed of three enzymes: pyruvate dehydrogenase (E1), dihydrolipoyl transacetylase (E2), and dihydrolipoyl dehydrogenase (E3), along with several cofactors, including thiamine pyrophosphate (TPP), lipoic acid, CoA, NAD+, and FAD. The reaction catalyzed by PDC involves the decarboxylation of pyruvate, the transfer of the acetyl group to CoA, and the reduction of NAD+ to NADH, resulting in the production of acetyl-CoA, CO2, and NADH.
The acetyl-CoA produced by PDC is a central metabolite that enters the citric acid cycle, where it is further oxidized to produce energy. The regulation of PDC is essential for controlling the flow of carbon into the citric acid cycle and is influenced by cellular energy status.
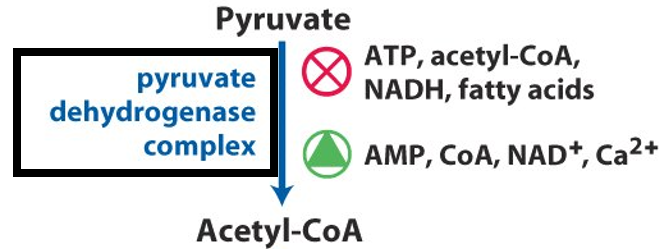
Pyruvate dehydrogenase complex is tightly regulated to ensure that pyruvate is converted to acetyl-CoA only when energy production is needed. The enzyme complex is regulated by covalent modification, specifically through phosphorylation and dephosphorylation.
Phosphorylation of PDC by pyruvate dehydrogenase kinase (PDK) inhibits the enzyme’s activity. PDK is activated by high levels of ATP, acetyl-CoA, and NADH, which signal that the cell has sufficient energy and does not need further acetyl-CoA production. Conversely, pyruvate and ADP activate the phosphatase enzyme that dephosphorylates PDC, thereby reactivating the enzyme and promoting the conversion of pyruvate to acetyl-CoA when energy is low.
In addition to covalent modification, allosteric regulation also plays a key role in the control of PDC. High concentrations of NADH and acetyl-CoA inhibit the enzyme complex, signaling that the citric acid cycle is running efficiently and that more acetyl-CoA is not required. On the other hand, ADP and pyruvate act as activators, indicating the cell’s need for more acetyl-CoA to produce ATP.
This regulation ensures that the conversion of pyruvate to acetyl-CoA is finely tuned to the cell’s metabolic needs, allowing the pyruvate dehydrogenase complex to operate efficiently under varying energy conditions.


