Pyruvate Carboxylase
Carbohydrate Metabolism
Pyruvate carboxylase catalyzes the conversion of pyruvate to oxaloacetate, playing a key role in gluconeogenesis.
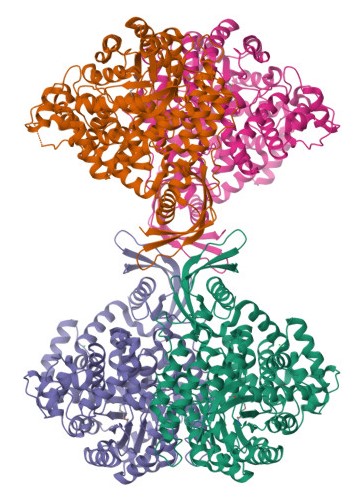
Pyruvate carboxylase catalyzes the conversion of pyruvate to oxaloacetate, a critical step in gluconeogenesis. This enzyme is primarily found in the mitochondria of liver and kidney cells, where it plays a pivotal role in maintaining blood glucose levels during fasting. Pyruvate carboxylase utilizes biotin as a cofactor and requires ATP to drive the carboxylation of pyruvate, adding a carbon dioxide molecule to form oxaloacetate. This step is essential because oxaloacetate is a key intermediate in both the citric acid cycle and gluconeogenesis.
In gluconeogenesis, oxaloacetate is then converted into phosphoenolpyruvate (PEP) by the enzyme PEP carboxykinase (PEPCK), which is a crucial step in the synthesis of glucose from non-carbohydrate precursors like lactate, glycerol, and amino acids. Pyruvate carboxylase, therefore, plays a crucial role in generating the precursors required for glucose production, especially during periods of fasting or prolonged exercise when glucose from dietary sources is not available.
In addition to its role in gluconeogenesis, pyruvate carboxylase also plays a critical role in the citric acid cycle. It replenishes oxaloacetate in the cycle, ensuring that the cycle can continue to operate and generate ATP and reducing equivalents for energy production. This is particularly important during times of high energy demand or when glucose is scarce.
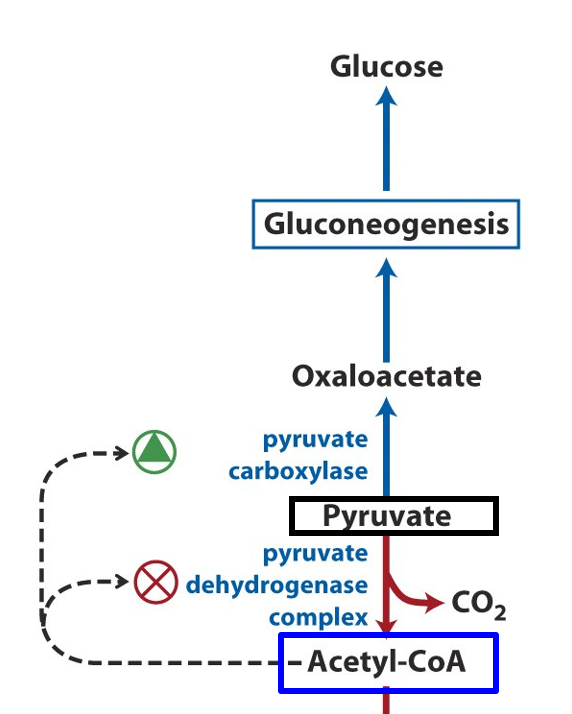
Pyruvate carboxylase activity is tightly regulated by several factors, ensuring that gluconeogenesis occurs only when necessary. The enzyme is activated by acetyl-CoA, which signals a need for more glucose production. When acetyl-CoA levels are high, it indicates that fatty acid oxidation is occurring and that there is a need to generate oxaloacetate for gluconeogenesis.
Conversely, pyruvate carboxylase activity is inhibited by high levels of ADP and AMP, which indicate low energy availability and signal the body to reduce glucose production. Additionally, the enzyme is subject to allosteric regulation, where glucose and insulin can influence its activity indirectly by modulating the enzymes involved in the overall gluconeogenesis pathway.
During fasting or starvation, when glucose is needed, glucagon and cortisol stimulate pyruvate carboxylase activity to enhance gluconeogenesis. These hormones promote the conversion of non-carbohydrate precursors into glucose, ensuring an adequate supply of glucose for tissues that rely on it, such as the brain and red blood cells.
Thus, pyruvate carboxylase plays a central role in regulating glucose metabolism, and its activity is carefully coordinated with the body’s energy status to balance glucose production and consumption.


