PFK-1
Carbohydrate Metabolism
Phosphofructokinase-1 (PFK-1) is a key enzyme in glycolysis, regulating the conversion of fructose-6-phosphate to fructose-1,6-bisphosphate.
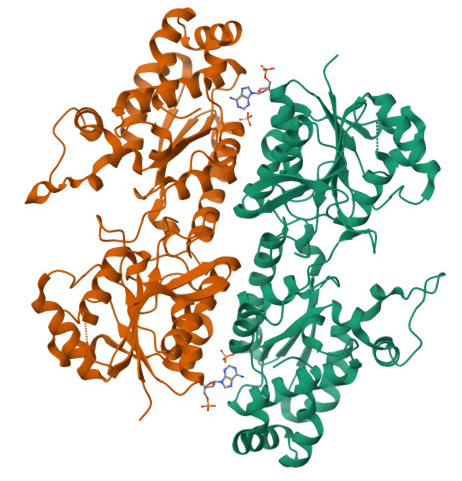
It is responsible for catalyzing the phosphorylation of fructose-6-phosphate to fructose-1,6-bisphosphate. This step is one of the rate-limiting reactions of glycolysis, making PFK-1 a central regulator of glucose metabolism. By committing glucose to glycolysis, PFK-1 controls the flow of carbon through the pathway and, in turn, influences the production of ATP, which is essential for cellular energy.
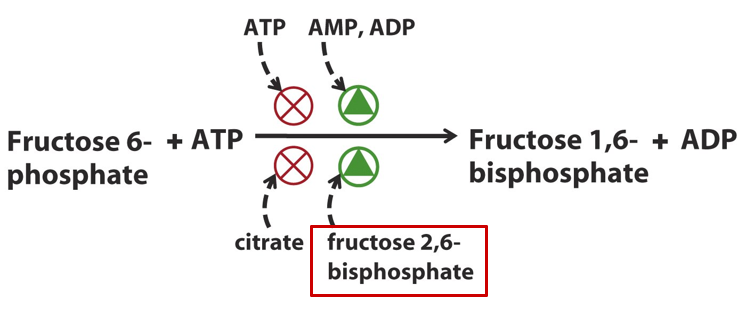
PFK-1’s activity is tightly regulated by cellular energy levels. When energy is low, AMP levels rise, activating PFK-1 and accelerating glycolysis to produce more ATP. Conversely, when ATP levels are high, PFK-1 is inhibited, preventing the unnecessary breakdown of glucose. Citrate, an intermediate in the citric acid cycle, also inhibits PFK-1, signaling that the cell has sufficient energy and reducing glycolytic flux. These regulatory mechanisms ensure that glycolysis is activated when energy is required and slowed when energy is abundant.
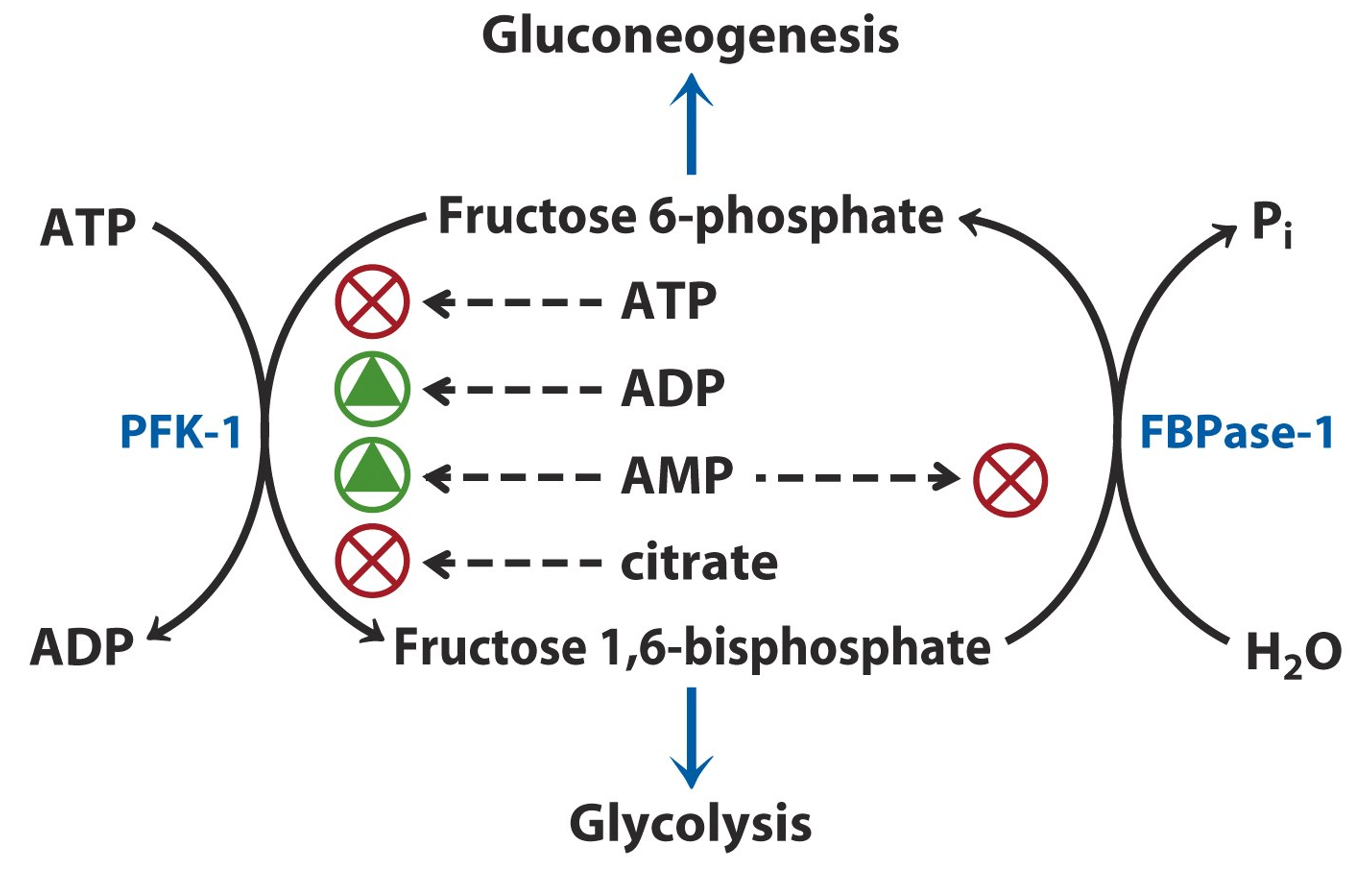
Beyond glycolysis, PFK-1 also plays a role in coordinating metabolic pathways. Its activity is influenced by hormonal signals such as insulin and glucagon. Insulin promotes the synthesis of fructose-2,6-bisphosphate, a potent activator of PFK-1, driving glycolysis when glucose is plentiful. On the other hand, glucagon, released during low blood sugar conditions, inhibits PFK-1 to favor gluconeogenesis, ensuring that glucose is conserved when energy is scarce.
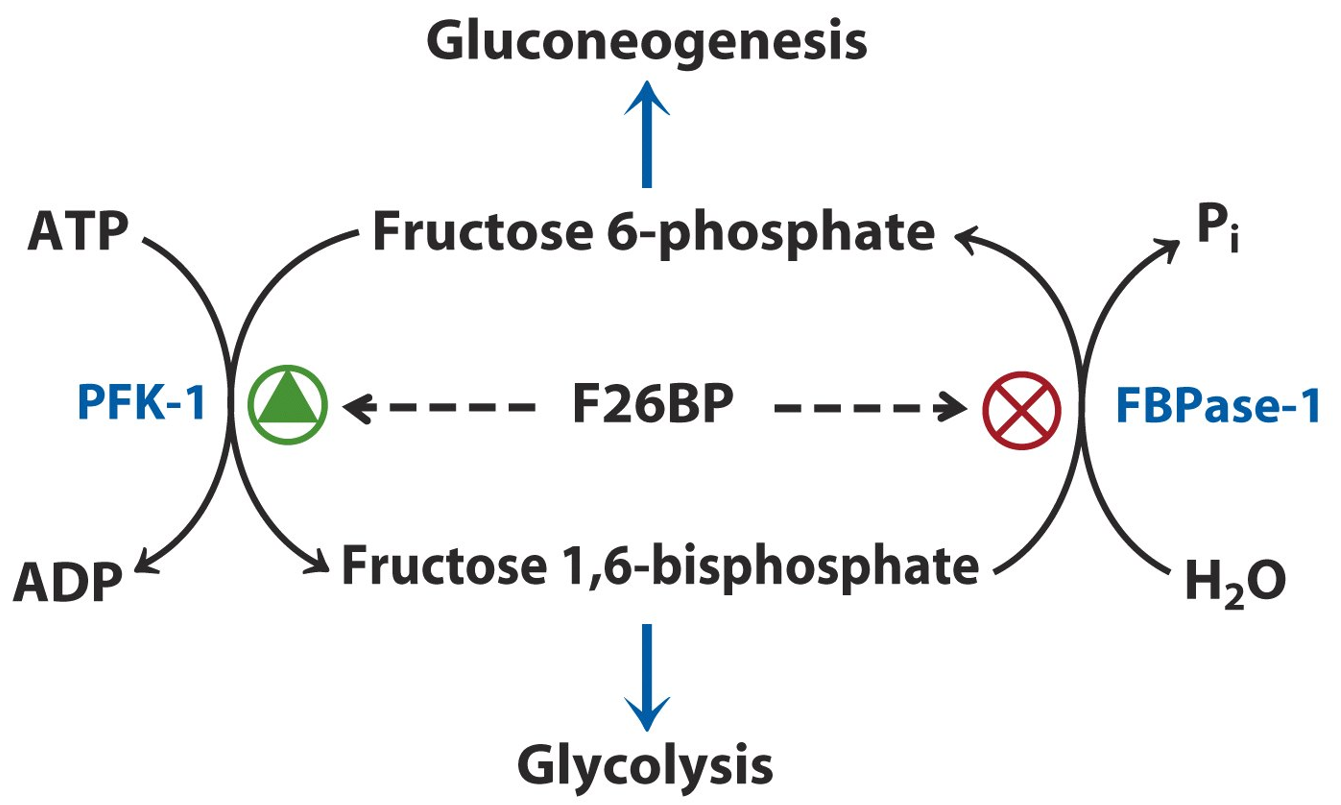
Thus, PFK-1 is integral to maintaining cellular energy balance and metabolic flexibility, ensuring that glucose metabolism is finely tuned to the cell’s energy demands and environmental signals.


