HMG-CoA Reductase
Lipid Metabolism
HMG-CoA reductase catalyzes the conversion of HMG-CoA to mevalonate, a key step in cholesterol biosynthesis.
HMG-CoA reductase catalyzes the conversion of HMG-CoA (3-hydroxy-3-methylglutaryl-CoA) to mevalonate, a crucial step in the biosynthesis of cholesterol and other isoprenoids. This enzyme is a central regulatory point in the cholesterol biosynthesis pathway, as it controls the rate-limiting step of the pathway. Mevalonate, the product of HMG-CoA reductase activity, is further metabolized into intermediates that lead to the synthesis of cholesterol, coenzyme Q10 (ubiquinone), and other essential biomolecules involved in cellular signaling, membrane structure, and protein prenylation.
HMG-CoA reductase is a membrane-bound enzyme located in the endoplasmic reticulum, where it is regulated by several mechanisms to maintain cholesterol homeostasis. The enzyme’s activity is influenced by the cellular cholesterol levels: when cholesterol levels are high, the enzyme is downregulated through a mechanism that involves feedback inhibition. Additionally, statins, a class of cholesterol-lowering drugs, inhibit HMG-CoA reductase, effectively reducing cholesterol synthesis in the liver and lowering blood cholesterol levels.
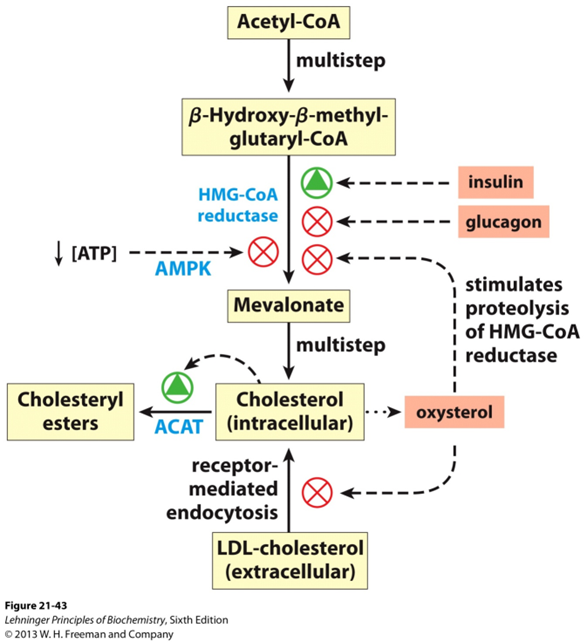
HMG-CoA reductase activity is tightly regulated to maintain cholesterol balance within the cell. The enzyme is regulated both by transcriptional control and by post-translational modifications, including phosphorylation and proteolysis.
At the transcriptional level, the expression of HMG-CoA reductase is controlled by the SREBP (sterol regulatory element-binding protein) pathway. When cellular cholesterol levels are low, SREBP is activated, leading to an increase in the transcription of HMG-CoA reductase, thereby enhancing cholesterol synthesis. Conversely, high cellular cholesterol levels inhibit SREBP, resulting in decreased expression of the enzyme.
Post-translationally, HMG-CoA reductase is regulated by phosphorylation. When cholesterol levels are high, AMPK (AMP-activated protein kinase) phosphorylates and inactivates HMG-CoA reductase, reducing its activity and thus decreasing cholesterol production. Additionally, ubiquitination of HMG-CoA reductase leads to its degradation in the proteasome, further lowering the enzyme levels and reducing cholesterol biosynthesis.
These regulatory mechanisms ensure that cholesterol synthesis is finely tuned to the needs of the cell, preventing the excessive accumulation of cholesterol while allowing for its production when needed.


