Glutamine Synthetase
Nucleic Acid Metabolism
Glutamine synthetase catalyzes the conversion of glutamate and ammonia to glutamine, playing a key role in nitrogen metabolism.
Glutamine synthetase catalyzes the conversion of glutamate and ammonia into glutamine, an important process in nitrogen metabolism. This enzyme plays a crucial role in maintaining nitrogen balance within the body by incorporating ammonia, a toxic byproduct of amino acid metabolism, into glutamine, a non-toxic compound that can safely transport nitrogen in the bloodstream. Glutamine is vital for a variety of cellular processes, including protein synthesis, nucleotide synthesis, and maintaining the acid-base balance.
The reaction catalyzed by glutamine synthetase involves the attachment of an amide group from ammonia to glutamate, forming glutamine. This reaction is ATP-dependent, and glutamine synthetase is widely expressed in various tissues, particularly in the brain, liver, and kidneys, where it is involved in detoxifying ammonia and maintaining nitrogen homeostasis.
Glutamine also serves as a precursor for the synthesis of other amino acids, such as proline, arginine, and glutamate, and is important for cellular metabolism, particularly in rapidly dividing cells like those of the immune system, intestines, and kidneys. It is also a key component of the urea cycle, where it helps to detoxify excess ammonia by facilitating its conversion into urea for excretion.
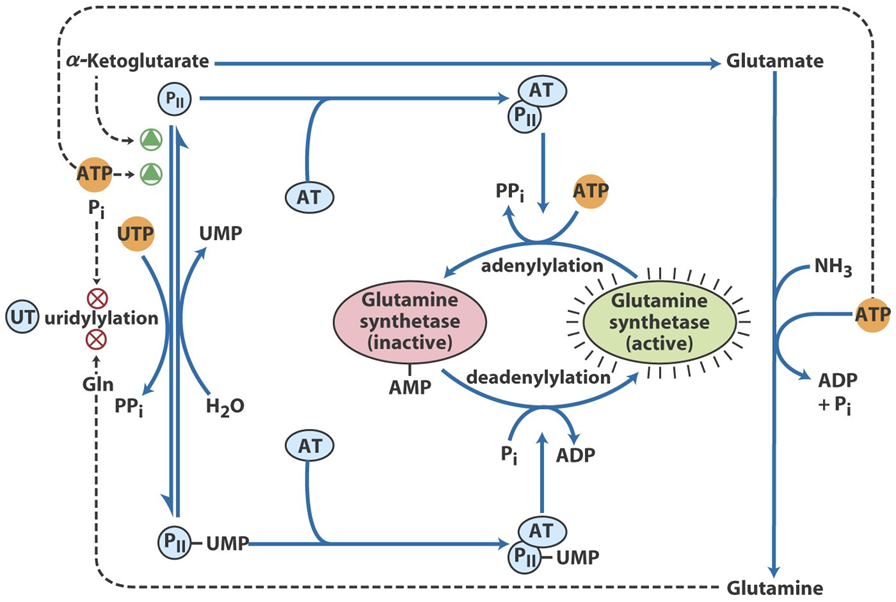
Glutamine synthetase is tightly regulated at both the transcriptional and post-translational levels to ensure efficient nitrogen metabolism. It is subject to allosteric regulation, where various end products of nitrogen metabolism, including glutamine itself, act as feedback inhibitors. High levels of glutamine inhibit the enzyme, preventing excessive synthesis of glutamine when the cell’s nitrogen needs are met.
In addition to this feedback inhibition, glutamine synthetase is regulated by covalent modification through a process called adenylylation, which involves the addition of an AMP molecule to the enzyme. Adenylylation reduces the activity of the enzyme, while de-adenylylation activates it. This covalent modification is modulated by adenylyltransferase, an enzyme that responds to changes in nitrogen availability in the cell.
Glutamine synthetase is also regulated by hormones, including glucocorticoids, which enhance its activity during stress, and insulin, which promotes its expression, particularly in muscle tissue. Through these various regulatory mechanisms, glutamine synthetase ensures that glutamine levels are tightly controlled, enabling efficient nitrogen metabolism and maintaining cellular and metabolic homeostasis.


