Carnitine Acyltransferase I
Lipid Metabolism
Carnitine acyltransferase I catalyzes the transfer of fatty acyl groups to carnitine, enabling fatty acid oxidation.
The enzyme catalyzes the transfer of fatty acyl groups from Coenzyme A (CoA) to carnitine, forming acylcarnitine. This modification is necessary because long-chain fatty acids cannot directly cross the mitochondrial membrane. By forming acylcarnitine, the fatty acid can be transported into the mitochondrial matrix, where it is then converted back to acyl-CoA by carnitine acyltransferase II (CAT II), enabling entry into the beta-oxidation pathway for ATP production.
The process begins when fatty acids are mobilized from adipose tissue and bound to CoA to form fatty acyl-CoA. However, since fatty acyl-CoA cannot pass through the mitochondrial membrane, it must first undergo a reaction catalyzed by carnitine acyltransferase I in the outer mitochondrial membrane. The acyl group is transferred from CoA to carnitine, forming acylcarnitine, which can then cross the membrane through a transporter. Once inside the mitochondrial matrix, the acyl group is transferred back to CoA by carnitine acyltransferase II, completing the transport cycle.
This transport mechanism is vital for the oxidation of long-chain fatty acids, a key metabolic process during periods of fasting, exercise, or starvation, when fatty acids are the primary energy source.
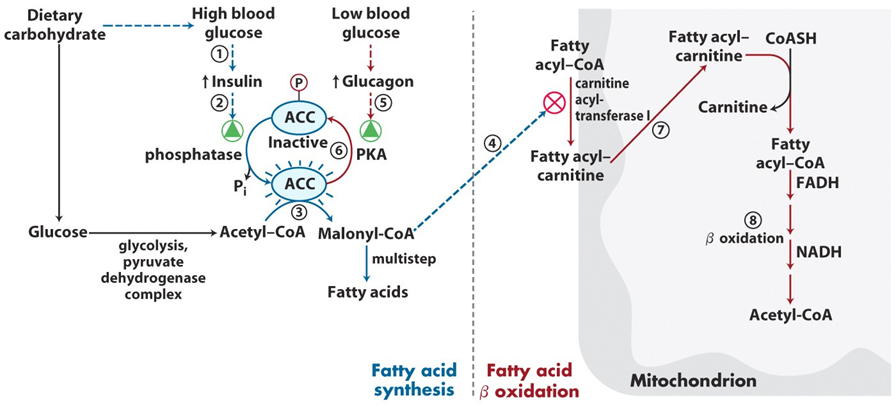
Carnitine acyltransferase I activity is regulated by several factors to ensure that fatty acid oxidation occurs appropriately based on the energy needs of the cell. Malonyl-CoA, an intermediate in fatty acid synthesis, acts as a potent inhibitor of CAT I. High levels of malonyl-CoA signal that the cell has sufficient energy and should not break down fatty acids, thus preventing unnecessary fatty acid oxidation and conserving energy for biosynthesis.
In contrast, when cellular energy levels are low, such as during fasting or exercise, AMP-activated protein kinase (AMPK) is activated and can indirectly promote the activity of carnitine acyltransferase I by decreasing the levels of malonyl-CoA. This allows fatty acid oxidation to proceed by lifting the inhibition on CAT I, facilitating the transport of fatty acids into the mitochondria for energy production.
Additionally, the activity of CAT I is influenced by the availability of carnitine. Adequate levels of carnitine are essential for the enzyme’s function, and carnitine supplementation can enhance fatty acid oxidation, particularly in muscle tissues. Therefore, the regulation of carnitine acyltransferase I ensures that fatty acids are transported and oxidized efficiently when energy demand increases, while avoiding unnecessary breakdown when energy stores are abundant.


