Acetyl-CoA Carboxylase
Lipid Metabolism
Acetyl-CoA carboxylase catalyzes the conversion of acetyl-CoA to malonyl-CoA, a key step in fatty acid synthesis.
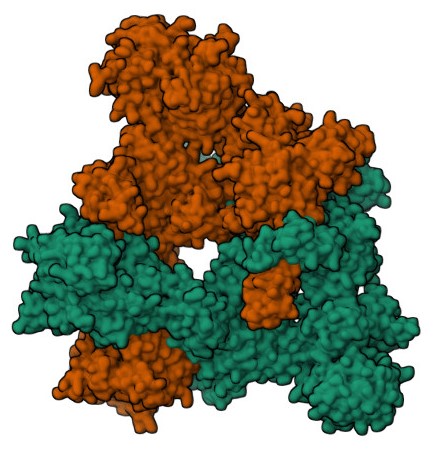
Acetyl-CoA carboxylase (ACC) catalyzes the conversion of acetyl-CoA to malonyl-CoA, a crucial step in fatty acid biosynthesis. This reaction is the committed step in the process of fatty acid synthesis and is highly regulated to ensure that fatty acid production occurs when needed, particularly during times of abundant energy. The enzyme requires biotin as a cofactor and operates by attaching a carboxyl group to acetyl-CoA, forming malonyl-CoA, which is then used in the elongation of fatty acid chains through the action of fatty acid synthase.
The formation of malonyl-CoA serves as both a building block for fatty acids and a key regulator of the process. Malonyl-CoA plays an essential role in regulating fatty acid oxidation as well. When malonyl-CoA levels are high, it inhibits carnitine palmitoyltransferase 1 (CPT1), which is necessary for the transport of fatty acids into the mitochondria for beta-oxidation. This inhibition ensures that fatty acids are not broken down while new fatty acids are being synthesized, allowing for a coordinated regulation of lipid metabolism.
Acetyl-CoA carboxylase operates in the cytoplasm, where it forms part of a complex regulatory network controlling both lipogenesis and lipid homeostasis. The activity of ACC is sensitive to the availability of its substrates, particularly acetyl-CoA, and is modulated by various metabolic signals.
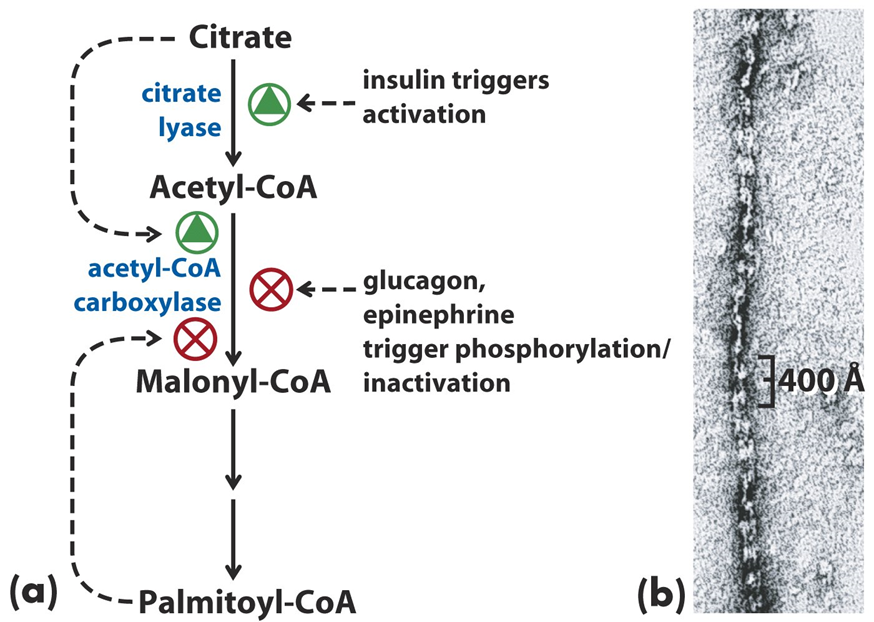
Acetyl-CoA carboxylase is regulated by a variety of mechanisms, including covalent modification, allosteric regulation, and hormonal signaling. One of the most important regulatory mechanisms is phosphorylation. ACC is inhibited by phosphorylation, which is mediated by AMP-activated protein kinase (AMPK) in response to low cellular energy levels (high AMP or low ATP), signaling the need to conserve energy. Phosphorylation inactivates ACC, preventing fatty acid synthesis when energy is scarce.
Conversely, ACC is activated by dephosphorylation, which is facilitated by protein phosphatase 2A and is promoted by high levels of insulin. Insulin, released in response to elevated glucose levels, signals a state of abundance and promotes the synthesis of fatty acids by activating ACC. Additionally, citrate, an intermediate of the citric acid cycle, acts as an allosteric activator of ACC. High citrate levels signal that the cell has sufficient energy and metabolites for fatty acid biosynthesis, further promoting the enzyme’s activity.
On the other hand, palmitoyl-CoA, a product of fatty acid synthesis, acts as a negative feedback inhibitor of ACC, preventing overproduction of fatty acids. This ensures that fatty acid synthesis is tightly controlled and balanced with the energy demands of the cell.
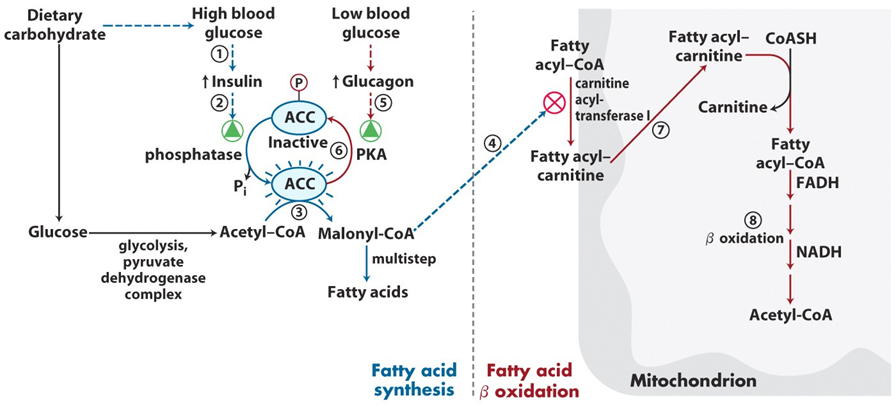
Through these complex regulatory mechanisms, acetyl-CoA carboxylase plays a central role in coordinating lipid metabolism, balancing the synthesis and oxidation of fatty acids in response to changing metabolic conditions.


